Carotid Artery
Applications of POCUS for the carotid artery and an introduction to spectral Doppler
The carotid artery is the first cardiovascular chapter because it is an ideal way to begin understanding spectral Doppler. However, this exam is also helpful for cardiovascular risk assessment and estimating patient fluid responsiveness.
CAROTID ATHEROSCLEROSIS
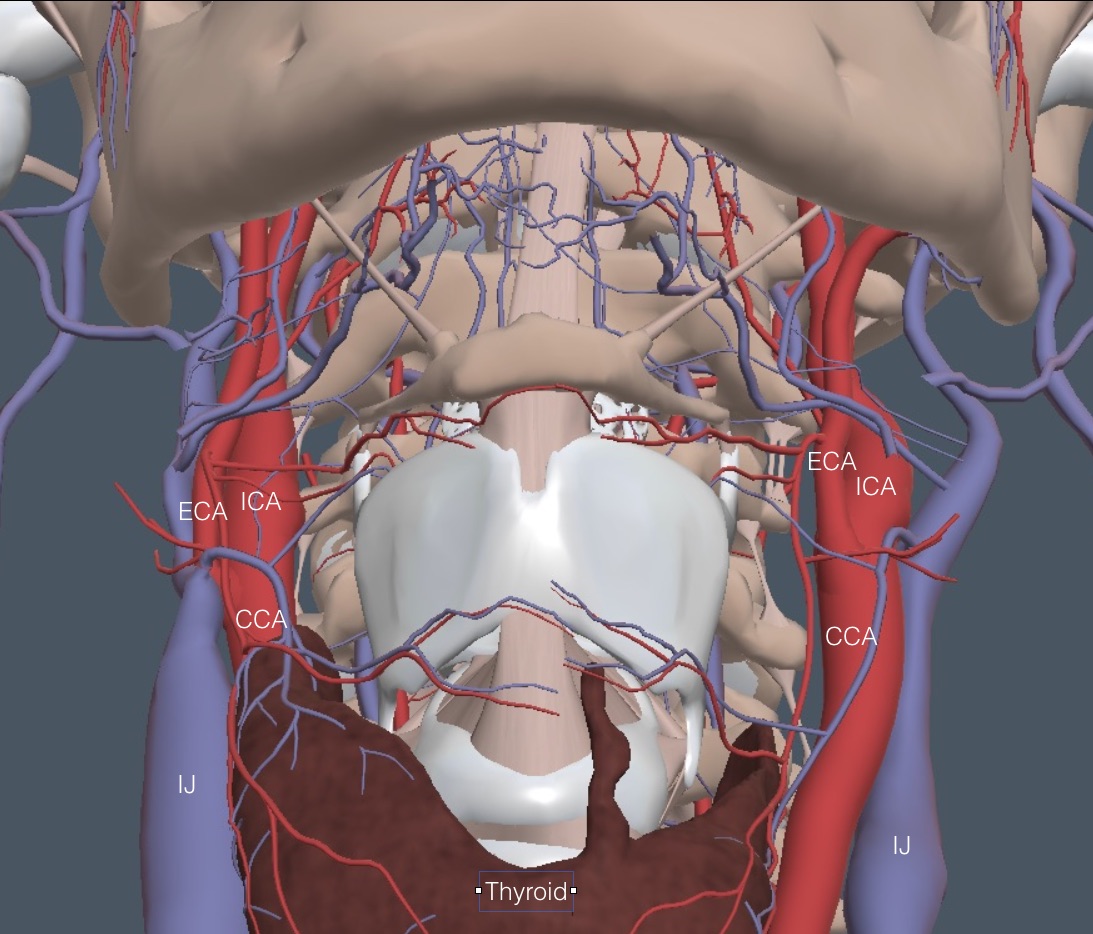
Carotid stenosis quantification is infrequently needed in the clinic (see section at the end of this chapter). However, detecting carotid atherosclerotic plaque is routine. Here is a depiction of the carotid artery anatomy in the neck. The internal carotid remains larger and more posterior than the external.
Technique: Have the patient supine. Plaque occurs near the bifurcation and into the internal and external carotids, with the internal carotid being more critical. Position the linear probe transversely in the mid-neck and center the short-axis carotid. Optimize the image and slide slowly to the bifurcation. If plaque is evident in the short-axis, estimate the amount of luminal narrowing. If plaque is equivocal in the short-axis, the long-axis often makes the plaque easier to see because the beam is perpendicular to more of the wall. The following clip shows an optimized short-axis right carotid at the beginning of the bifurcation. Non-obstructing hyperechoic plaque is seen.
Next is a moderate plaque in a longitudinal carotid at the beginning of the internal carotid. Stenosis was minimal with this plaque.
Carotid plaque confirms non-coronary atherosclerotic disease, placing the patient in a higher risk category. The more plaque the greater the risk. Patients at intermediate risk for atherosclerotic vascular disease may first undergo a carotid plaque survey instead of a coronary CT calcium study (CAC). Carotid plaque is strongly associated with CAC, but it is partly independent, such that “individuals with carotid plaque, despite the absence of CAC, might benefit from more intensive primary cardiovascular prevention efforts to help reduce cardiovascular risk.” (Circulation: Cardiovascular Imaging 2021: https://doi.org/10.1161/CIRCIMAGING.120.011701)
AN EXERCISE IN SPECTRAL DOPPLER
Ultrasound Doppler includes Color Flow Doppler (Color), Power Doppler Imaging (PDI), Pulse Wave Doppler (PW), Continuous Wave Doppler (CW), and Tissue Doppler Imaging (TDI). Color and PW will be explored in this chapter and are essential for some cardiovascular exams. PDI, CW, and TDI will be introduced in other chapters.
Image the long-axis right common carotid and optimize the view.
Activate Color flow Doppler: The default Color sector box width and height for the carotid setting on Venue are appropriate for most carotids. The greater the width of the box, the lower the frame rate for the Color image. The height of the box does not affect the frame rate, but excessive height can lower the scale of the Color flow, resulting in greater aliasing (discussed in detail in several heart chapters). Optimizing the width and avoiding excessive height of the Color sector box applies to any use of Color. The default carotid Color sector box is tilted +20 to enhance the display of red flow coming from the heart on the right. Adjust the GAIN so Color fills the vessel without speckling (flash) beyond the artery. Here is a clip of optimized Color in a long-axis common carotid.
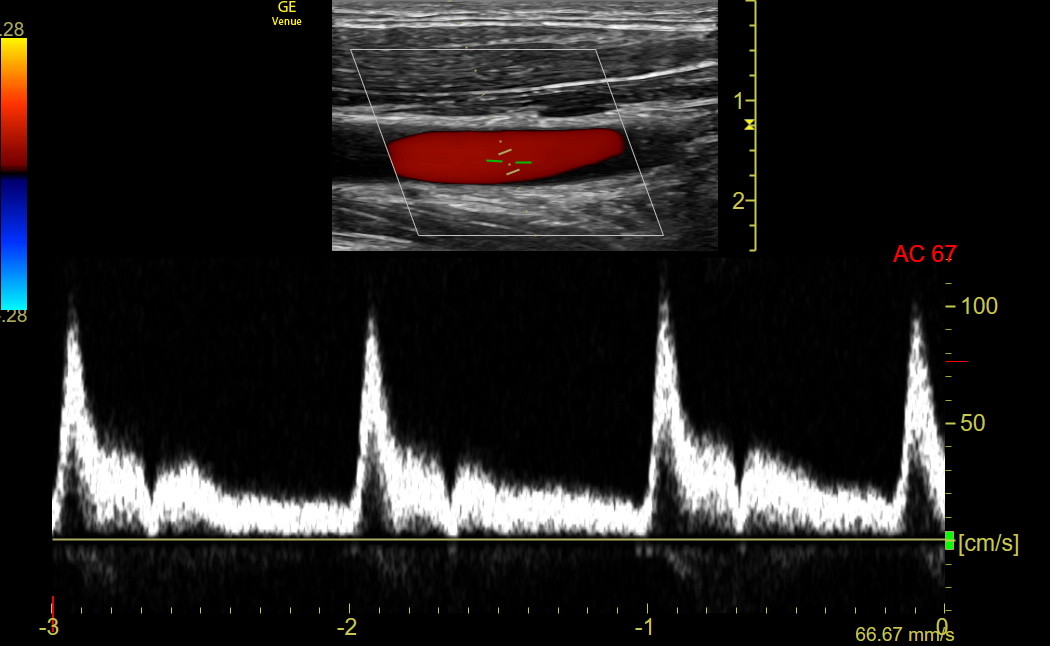
Activate PW: PW measures blood flow velocities in the gate (between the two hash marks) and is limited to about 2.5 m/sec. Move the gate to the middle of the artery and use Angle Correct to rotate the line parallel to flow so velocity is not underestimated. The following is an optimized PW tracing showing appropriate gate placement in the upper B-mode image.
Make sure the PW complexes occupy most of the graph. The Baseline can be moved with a finger, and the complex size can be adjusted by vertically contracting and expanding the left thumb and index finger. The tracing above is excellent. The peak velocity of a complex could have been measured and would have been about 1.0 m/sec.
Changing the sweep speed: The sweep speed is the recording speed. A fast sweep speed spreads things out, showing fewer complexes but greater detail of individual complexes. A slow sweep speed crunches complexes together to see many without detail. The carotid flow time discussed next needs a medium or fast sweep speed. However, a slow sweep speed is required to assess carotid velocity variation with respiration, as discussed in the chapter on Pericardial Tamponade. On Venue, the sweep speed for any spectral Doppler tracing is easily changed on the horizontal time axis with a pinch or expansion of the thumb and index finger.
CAROTID FLOW TIME (CFT)
The carotid flow time measures LV ejection time, a classical measure of LV function dependent on heart rate, preload, afterload, and contractile state. Like the Velocity Time Integral (VTI, discussed later in the Left Ventricle chapter), the CFT reflects the left ventricular outflow. It is optimal to capture the CFT at end-expiration when left heart volume and function are maximal. The sweep speed should be adjusted so only 3-4 complexes show on the screen. The complex analyzed must show a dicrotic notch indicating aortic valve closure. The second complex in the tracing above is excellent for measurement.
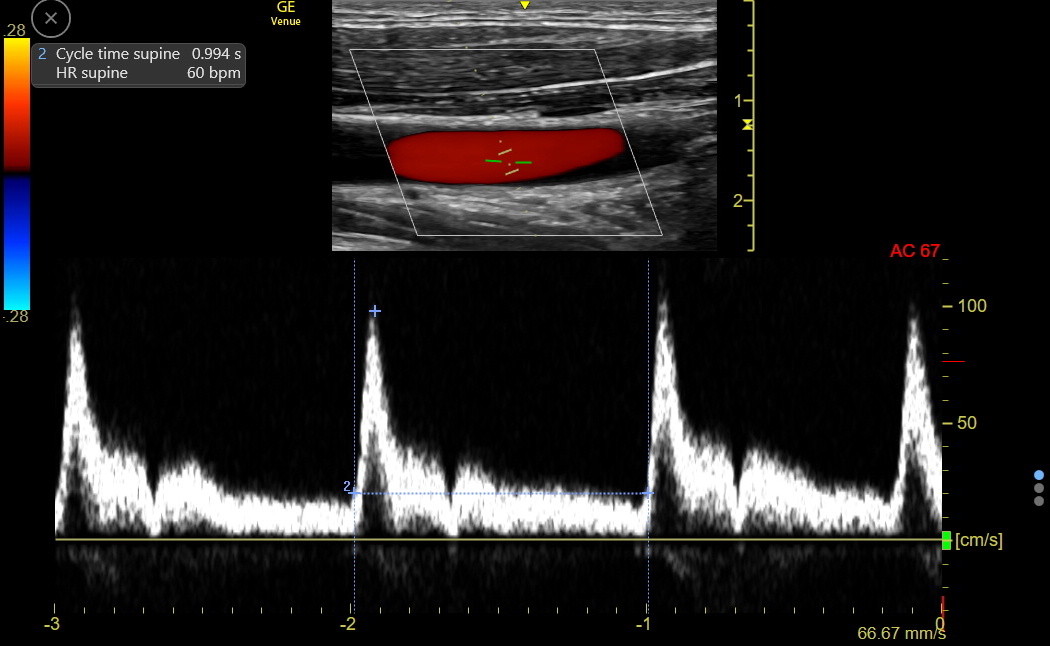
Measure the Cycle Time: put a first caliper at the start of the carotid upstroke and an ending caliper at the beginning of the next upstroke. This is the CYCLE TIME (CT). In the following tracing, the CT was 0.99 seconds. Venue calculated the heart rate (HR) as 60/0.99 = 60.6 bpm but rounded down.
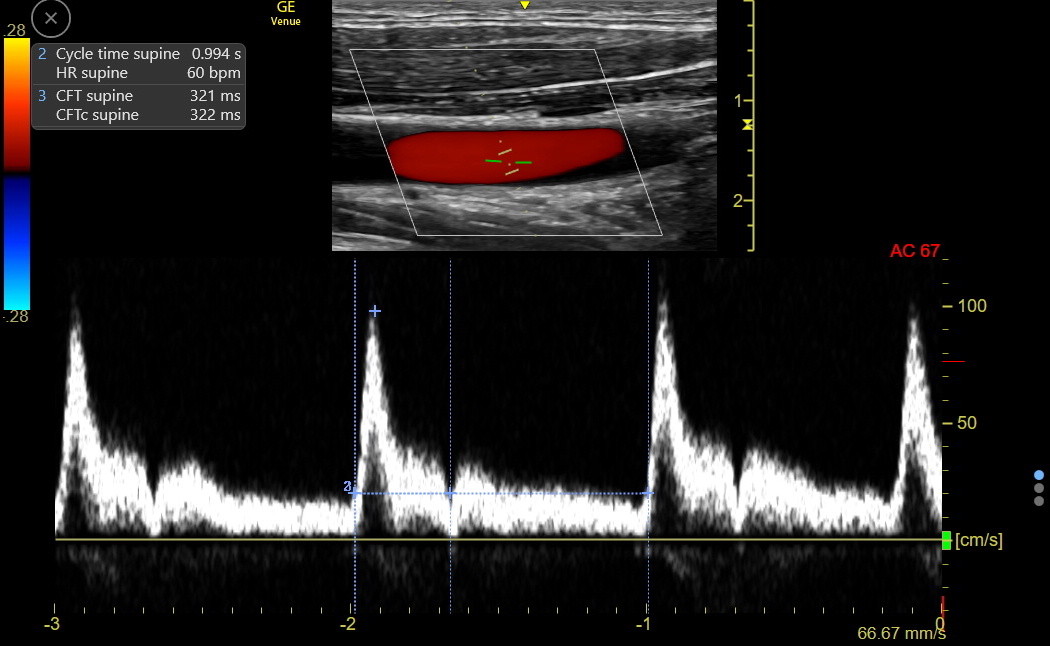
Measure the Carotid Flow Time (CFT): Put the first caliper again at the start of the carotid upstroke and measure to the central depth of the dicrotic notch. This is the raw CFT, which was 321 msec in this example.
Venue corrects the raw CFT to a heart rate of 60 bpm using an empirically derived formula:
Corrected CFT (CFTc) = raw CFT + [1.29 x (HR – 60)]
This patient’s CFTc was 322, virtually unchanged from the raw CFT because the heart rate was barely over 60 bpm..
A series of 142 normal volunteers showed a 95% normal range of about 280 – 360 for the supine CFTc. Notably, a 45-degree passive leg raise for 1 minute caused a mean change of about 2% in the CFTc in these normal adults, although a few showed more significant changes because the CFTc is sensitive to changes in volume status. The leg raise requires a third person to lift the legs; patients must not help with the elevation, or the associated Valsalva maneuver distorts the results. This two-part procedure is called a dynamic CFTc.
Many critical care physicians distinguish “fluid tolerant” from “fluid responsive.” A fluid-tolerant patient can receive fluid without immediate harm, but the cardiac output may or may not increase. A fluid-responsive patient shows increased cardiac output with fluid; fluid tolerance may or may not be present. The dynamic CFTc detects fluid responsiveness, not fluid tolerance. As the increase is progressively greater than 5%, the evidence for fluid responsiveness strengthens. A small ICU study did passive leg raise on patients with an indwelling pulmonary artery catheter (PAC). The change in cardiac output was compared to the change in the CFTc. A CFTc increase of 9% had a sensitivity and specificity of about 90% for an increase in cardiac output (Jelic, POCUS J, 2023).
CAROTID STENOSIS: For the Advanced IMBUS physician, determining the degree of carotid stenosis is like assessing the severity of aortic stenosis. The peak PW velocity in the stenosis is measured and compared to the velocity in the mid-common carotid. The pre-and post-stenosis velocity ratio is interpreted like the dimensionless index (DI) for aortic stenosis, except that the numerator and denominator are reversed for carotid stenosis. Peak velocity through a stenosis less than 1.25 m/sec with a ratio less than 2.0 compared to the common carotid indicates stenosis less than 50%. Peak velocity of 1.25 – 2.3 m/sec and a ratio of 2.0 – 4.0 is stenosis from 50 - 70%. Peak velocity greater than 2.3 m/sec with a ratio greater than 4.0 indicates more than 70% stenosis. Nearly occluded carotids are probably a visual diagnosis because only very narrow residual flow may be seen, and getting the PW gate in the small residual jet is difficult.